Flexible and protected – New findings on SARS-CoV-2 protein shed light on virus’ ability to infect cells.
Sars-CoV-2 protein shed light on virus’ ability to infect cells
In the fight against the coronavirus SARS-CoV-2 researchers from various research institutions in Germany joined forces to study the spike protein on the surface of the virus – this protein allows the virus to bind to and to infect human cells. The study shows surprising insights on the spike protein, including an unexpected freedom of movement and a protective coat, hiding it from antibodies. The results are published in Science
To bind to and to infect human cells the coronavirus SARS-CoV-2 requires a special protein on its surface. These spike-like structures are at the centre of vaccine development because they trigger an immune response in humans. Therefore, a group of German scientists, including members from the Max-Planck Institute of Biophysics, EMBL Heidelberg and the Paul Ehrlich Institute focussed their studies on the surface structure of the virus to gain insights for the development of vaccines and effective therapeutics to treat infected patients. Knowledge of the spatial structures are central to understand the individual steps of the infection process, such as penetration into the target cells.
The team combined technologies of cryo-electron tomography, subtomogram averaging and molecular dynamics simulations to manage the analysis of molecular structures of these spike proteins in their natural environment and with near-atomic resolution. 266 cryo tomograms of about 1000 different viruses were generated, each carrying an average of 40 spikes on its surface. Subtomogram averaging and image processing finally provided the needed important structural information from these spikes.
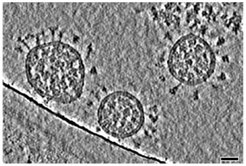
The results were surprising: The data showed that the globular portion of the spike protein, which contains the receptor-binding region and the components necessary for fusion with the target cell, is connected to a flexible stalk. “The upper spherical part of the spike has a structure which is well reproduced by recombinant proteins used for vaccine development,” explains Martin Beck, Group Leader at EMBL and Director at the MPI of Biophysics. “However, findings about the stalk, which fixes the globular part of the spike protein to the virus surface, were new.”
“Little was previously known about this structure,” adds Gerhard Hummer, from the MPI for Biophysics and the Institute of Biophysics at the Goethe University Frankfurt. “Looking at our computer models and the actual images, we discovered that the stalks are extremely flexible. They rarely stand straight on the membrane, but are inclined in all directions.” The team identified four different domains in the stem, which they called the hip, knee, ankle, and finally the foot section, which is embedded in the membrane. By combining molecular dynamics simulations and cryo-tomography, they demonstrated that these domains can perform bending movements, explaining the flexibility of the spikes.
SARS-CoV-2 spike proteins
“Like a balloon on a string, the spikes appear to move on the surface of the virus and thus are able to search for the receptor for docking to the target cell,” describes Jacomine Krijnse Locker, group leader at the Paul Ehrlich Institute, the results. The analyses finally showed that the stalk is equipped with many glycan chains. These glycan chains might also provide the stalk with a kind of protective coat, thereby hiding it from neutralising antibodies. Another important finding on the way to effective vaccines and medicines.
Source Article
Beata Turoňová et al. In situ structural analysis of SARS-CoV-2 spike reveals flexibility mediated by three hinges Science, published on 18 August 2020
DOI: 10.1126/science.abd5223

